|
AIR POLLUTION and THE WORLD'S CLIMATE (University of Adelaide, 1984) [Amended with a few corrections] Anonymous (Investigator #196, 2021 January) [37 years after writing this essay in 1984 the author
wrote two updates on the reality of climate change — here.] Abstract
A review of air pollution on global climate is presented. The
potentially climate-affecting air pollutants include carbon dioxide,
chlorofluorocarbons, methane, carbon monoxide, sulphur dioxide,
nitrogenous gases, particulates and heat. Some pollutants trap infrared
and promote a greenhouse effect. Others may promote cooling by
increasing cloud and surface reflectivity. The air pollutants interact
with each other and with surface pollution, biomass, ice caps, oceans,
solar radiation and other determinants of climate. A logical problem
arises from our inadequate definition of "normal climate".
1. Introduction
Air pollution is the introduction of substances or heat into the air as
a result of man's activities in such quantities as to be measurable
over wide areas. The meaning of "wide" is subject to context. Since I
am dealing with "world" climate, "wide" will in the case of most
pollutants mean "worldwide". Carbon dioxide (CO2),
though a substance occurring naturally in the air, is a pollutant when
man's activities measurably increase its concentration worldwide.
Emissions not of human origin, such as volcanic emissions, are by
definition not "pollution".
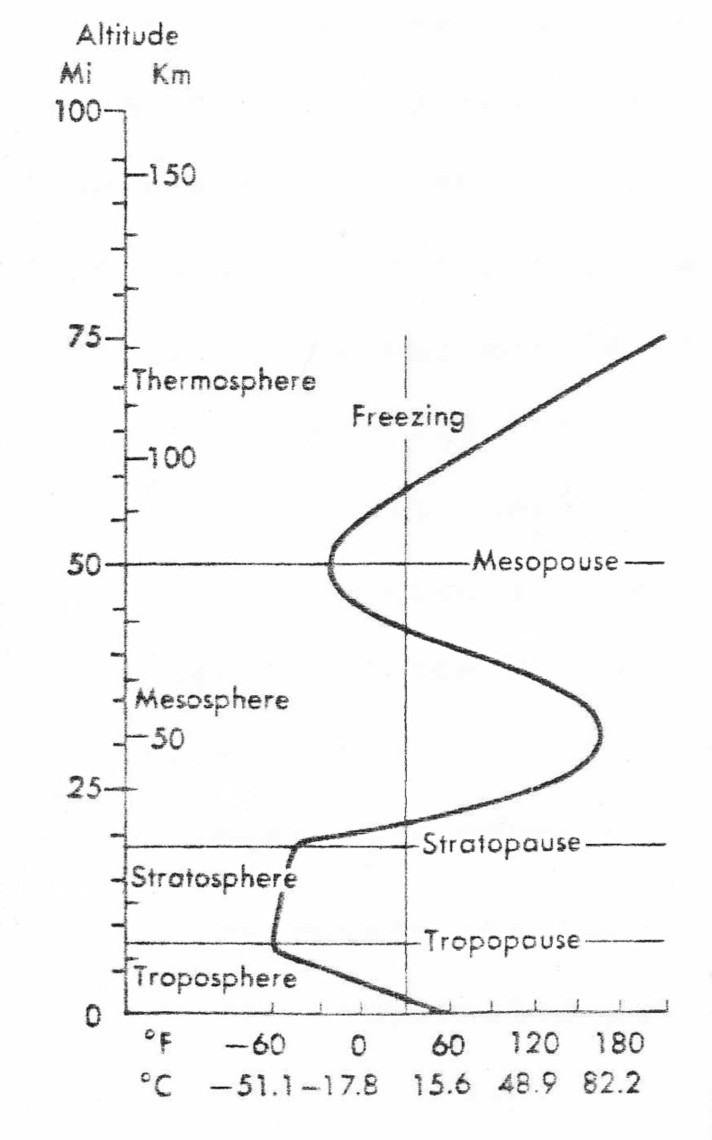
"Weather" refers to short-term (usually weekly or less) local conditions of wind, temperatures, cloudiness and precipitation. "Climate" refers to daily, weekly, monthly, seasonal or yearly averages or ranges calculated over long periods. "Normal climate" is internationally defined as the conditions prevailing 1931-1960 (Bolin, 1977). Climatic-change would be evident if averages changed, variability changed or both changed. Changed variability would be manifested in "extremes" of weather. Climate results from the interaction of processes involving solar radiation, the atmosphere, land, oceans, biota, reflectivity, infrared radiation, and clouds. My emphasis will be on the atmosphere. But where air pollution threatens climatic change indirectly by influencing Earth's surface such as the biota, ice caps or oceans, the latter will need to be mentioned. The temperature of the atmosphere varies with altitude (Figure 1). Our concern in this essay is with climate that humans experience, therefore concerned with the lower levels, mainly the Troposphere. Figure 1 (From: Ramsey et al 1979)
 Pollutants (Table 1) are added to the air by three major processes:
Attrition is the addition of pollution to air via friction such as
asbestos from break linings, carbon from tyres and when industry
grinds, blasts or cuts. Vaporization occurs when liquids turn to gases
and diffuse into the atmosphere. Combustion is burning and the chief
pollutants in this process come from car engines.
Table 1
World total primary pollution estimates.
From Greenwood and Edwards (1973)
Air pollution may alter the effects of other climate-influencing
variables. It is also necessary to distinguish natural fluctuations or
"noise" in these different climate-affecting factors from the effects
pollution may have.
A controlled scientific experiment to investigate air pollution is not possible since we have but one Earth. We can however compare regional climates before and after pollution. We could also compare two or more areas of similar initial climate when one area experiences marked air pollution and the other does not. The results can only be suggestive since the procedure does not allow for the interactions of the factors mentioned above. The similar climates of two areas may result from different interactions between oceans, mountains, air movements, clouds, etc. 2. Models
A partial solution is the use of models. Models can be actual scale
built physical models of continents, mountain ranges, etc. Wind tunnels
and fans can simulate winds, and artificial heat can mimic solar heat.
Early studies of the general atmospheric circulation used a 1 metre
radius pan containing 5 cm of water, heated at the rim and cooled at
the centre to simulate equator and poles (Stringer, 1972). Observations
on models are compared to data gathered in the real world, and new
models are improved accordingly.
Nowadays there are mathematical models — really sets of complex mathematical formulae – through which computers simulate large-scale climatic events using the laws of physics, chemistry and thermodynamics. The model is essentially a set of equations, which the computer solves. A number of atmosphere-simulating models were described by Fels et al (1980) and features of atmosphere/ocean models by Kellogg and Schware (1981). Atmosphere and ocean models are used successfully in weather forecasting, explanations of past climates and simulations of current annual climatic cycles. Manabe and Stauffer (1981) used current CO2 levels for the control run of a climate model and showed that the model successfully reproduced large scale aspects of seasonal and geographic atmospheric structure. These successes provide grounds for confidence in forecasts of the effects of pollution on climate. Of course the computer's answer will be subject to the initial assumptions and input. These are continually being refined and updated. The acid test of a model is when it not only simulates current climates accurately, including the summer winter differences (Chervin, 1980), but its forecasts correspond to data collected in the future. Then, using inductive logic we might expect that forecasts still further future are also reliable. Whether or not induction is the process by which science progresses, is debated. (Chalmers, 1976; Swinburne, 1974). In our topic, particular inductive generalizations will be counted as "good" until they are falsified. 3. Carbon Dioxide
Descriptions of the carbon cycle, the processes that maintain the atmospheric CO2 concentration, are available in basic geology or biology textbooks such as Ramsey (1979) and Hardin (1961).
According to Ramsey the processes that increase and decrease atmospheric CO2 are: Increase (Millions of Tons):
• Respiration, decay (60) • Combustion of fossil fuels (6) • From soil (2) • Volcanism (0.1) Decrease (Millions of Tons): • Photosynthesis (60) • Weathering of rocks (0.1) • New fossil material ( <0.1
The only component perhaps out of balance is the one involving fossil fuels — the atmospheric CO2 from this is currently rising (Table 2).
Table 2
Atmospheric CO2 concentrations in parts per million From Keeling (1978) and Pittock et al (1981)
Deevey 1958) attributed most of the increase to the drying of bogs and
marshes. The steepening rise since then, paralleling man's increased
emission (Table 3) renders his conclusion unlikely. The burning of
coal, petroleum and vegetation is increasing. About 1/6 of the world's
fuel use consists of wood and half of all trees cut down are used for
heating and cooking (Hall, 1979).
Table 3
Fraction of carbon emissions (%) by developed and developing countries. From Kellogg and Schware (1981)
John Tyndall in 1861, the Swedish chemist Svante in the 1880s, Callender (1938), and Plass (1956) warned of a CO2
greenhouse effect. Global warming before-WW2, together with
precipitation changes (Budyko 1977), seemed to confirm the hypothesis.
The actual warming mechanism is as follows:
CO2 is transparent to solar radiation at visible wave-lengths which therefore reach Earth's surface unimpeded by CO2.
Outgoing long-wave (infrared) radiation tends to be absorbed and
re-radiated. Increased gas concentration (including ozone and water
vapour discussed later) will therefore elevate thermal and radiative
fluxes until a new balance is reached with incoming radiation.
Manabe et al (1975) modelled the climatic effect of CO2. Climate models are first given a control run to establish how their "climate" compares to the real world. Then the model is run again with increased (doubled or quadrupled) CO2. This is the "perturbed run". The result in the 1975 experiment was a 2.9oC warming. The model used was however overly simplistic and incorporated assumptions of constant cloudiness in different latitudinal zones, idealized geography and no heat transport by ocean currents. The results from other models for CO2 doubling vary from 2oC temperature rise (Manabe and Stouffer, 1979) to about 4oC. Manabe and Weatherald (1980) modelled the more sophisticated situation of climatic response resulting from interaction between CO2, radiation and cloud cover. The model response was a 2.4oC increase, pole-ward transfer of moisture and latent heat, pole-ward retreat of highly reflective snow and reduction of temperature gradients in the lower troposphere. Evaporation rate increased uniformly in all latitudes but the mean precipitation rates were latitudinally dependent. The mid-latitude (35o – 45o North) rain-belts shifted pole-ward increasing the continental run-off in high latitudes and decreasing it in mid latitudes. The model assumed a single mountain-free continent. The atmospheric-ocean coupling was represented unrealistically and therefore ocean current heat transport heat exchange between surface and deep waters were not represented. Cloud-response effects were also ignored. Another model study (Manabe et al, 1981) revealed mid and high latitude soil moisture decreases due to briefer snowmelt seasons and a correspondingly longer warm season. Mid-latitude rain belts shifted pole-ward and the spring rainfall reduction commenced earlier. The summer dryness suppressed evaporative heat-loss leading to further continental surface heating in areas of reduced moisture. A flaw in all these models is that only the model's response from a state of climatic equilibrium was considered. The researcher would perturb the model with a sudden doubling of CO2 concentration and await a new equilibrium in the model climate. In the real world CO2 does not double instantaneously but changes in concentration gradually. The models also took little account of mechanisms which remove CO2 from the air (= sinks) — mechanisms which may work more effectively as the concentration rises. Most carbon now deposited was at one time in the atmosphere. Therefore sinks must exist. This also follows from our understanding of the carbon cycle. The current rate of removal is indicated by Revelle (1982). From 1959 to 1978 the atmospheric CO2 content increased by 4 x 1010 tons whereas the emission from human fuel and industry was 7 x 1010 tons. Bolin (1977) stated that the CO2 tonnage injected into the air annually was sufficient to increase the atmospheric concentration 2.3 ppm. The observed rise however was only 1.2 ppm annually. Croplands contain 1/10 to 1/20 the carbon per unit area that forests contain. Forest regrowth on abandoned agricultural land together with deliberate planting of forests such as China's "great green wall" may constitute partial compensation for fossil fuel burned. Pearman and Hyson (1981) discovered that the Northern Hemisphere CO2 concentration is not rising steadily but shows considerable fluctuation, with the amplitude of fluctuation having increased by 8.6% in the 1970s. By comparison with the Southern Hemisphere, the authors concluded that the oceans modulate the concentration by only 0.5 ppm. The major cause of the fluctuation was suggested to be the interplay between fossil fuel burning and an annual increase in net ecosystem production. Carbon stored in the seasonal biosphere increased in 1958-1978 by 0.52 x 1012 Kg. Seiler and Crutzen (1980) however were uncertain as to whether there was a net uptake or loss of carbon by the biosphere. The authors indicated doubt regarding whether their method, based largely on extrapolations from land use and demographic data, was valid. They did however indicate another CO2 sink, ignored by previous researchers, namely charcoal which decomposes very slowly. Furthermore Rosenberg (1981) predicted that CO2 increase of from 200 to 600 ppm will lead to significantly increased photosynthesis and hence in plant productivity in both forests and cultivated plants. This would slow down the CO2 increase. Rosenberg however was unable to confirm his hypothesis by actual CO2 seeding of fields because the CO2 was too readily dispersed by turbulence. Singer (1970) pointed out that the oceans contain 60 times as much CO2 as the atmosphere. However most oceanic CO2 is not in the form of free carbon dioxide but as carbonate and bicarbonate ions (Revelle, 1982). Hence a large change in the CO2 pressure produces a comparatively small change in free oceanic CO2. Oceanic absorption was in fact lower than previously supposed. Furthermore, ready atmospheric/oceanic gas exchange occurs only in the upper 80 metres of ocean and the CO2 absorbed in this layer changes only 1/9 as much as any atmospheric changes. Downward convection and diffusion are much slower processes and make even less difference. What oceanic absorption does occur might however result in increased oceanic photosynthesis. But little study has been done on this possibility. A new area of research was commenced by Hoffert et al (1980) who pointed out that the oceans might act as a thermal buffer and absorb CO2-trapped heat. Surface winds cause thorough mixing down to 50 metres and this could delay global surface temperature response by 5-20 years (Thompson and Schneider 1981; Cess and Goldenberg, 1981). Slower heating occurs to the deepest depths. This is another effect ignored in previous models. The Hoffert et al model simulated global oceanic heat capacity in an ocean model with specified upwelling and vertical diffusion. Results indicated a 10-20 years lag in CO2-induced temperature response by the year 2000. Oceanic warming may also change the circulation patterns of ocean currents (Hartline, 1980). This would in turn alter the climates of countries adjacent to the currents. If global warming fails to occur at the predicted rate this does not falsify the greenhouse hypothesis but shows that it was overly simplified. The ocean's influence extends further. There is much regional variation in its mixing. The polar response is poorly understood. Down welling polar seas produce much of the connection between surface and deep waters. Further regional variation in oceanic heat mixing is due to the geographic distribution of oceanic heat capacity. Hence thermal time lags will vary regionally and this will make any unambiguous detection of global warming difficult. Berner et al (1980) and Delmas et al (1980) analysed trapped air in polar ice cores and demonstrated wide atmospheric CO2 variation in the past 30,000 years. In 3000 BP for example the concentration was 30% greater than the current 340 ppm and 12,000 BP it was only 200 ppm. The causes of the variation are obscure. Estimates from climate models, however, suggest that such CO2 variation represents a temperature variation of only 4oC whereas the variation between the glacial ages would have been about 8oC. In other words, CO2 change was only one mechanism in past climatic change (Thompson and Schneider, 1981). Furthermore, CO2 variation irrespective of man's activities is wider then hitherto suspected. This does not necessarily imply that CO2 pollution by man is irrelevant. If an 8oC swing represents the difference between glaciation and warmth, man's potential 2oC via CO2 influence may decisively moderate or exacerbate other trends. Could a warming trend be detected by monitoring changes in ice cap volumes in conjunction with changes in ocean levels? The discharge rate of Antarctic ice depends on the horizontal pressure gradient which itself depends on the thickness of the ice which in turn is related to precipitation rates. Oerlemans (1982a) used a simple model for Antarctic ice flow. It incorporated a rate equation for ice thickness, precipitation variability estimates obtained via ice cores, and an additive stochastic term to represent accumulation rate fluctuations. By estimating the time intervals required for returns to equilibrium Oerlemans concluded that sea level variations of 4 cm per century should not be unusual and that this "noise" is inseparable from secular trends being sought. From another model study Oerlemans (1982b) concluded that CO2 climatic warming would raise polar precipitation on the Antarctic ice sheet, lowering the ocean levels. This is the reverse of previous commonsense expectations! The model ignored the possible response of the Greenland ice sheet and the effects of possible Antarctic ice sheet instability due to snow accumulation. The largest ice volume increases, of 0.5%, resulted from small temperature increases of 2oC. An estimated 0.3 metre ocean level drop in 200 years was suggested. All the studies cited thus far ignored the obvious possibility that increased atmospheric water vapour will follow from even slight warming. With the lower atmosphere warmed, clouds will form at higher altitudes than previously. The clouds will then emit less infrared radiation to Space — assuming that the rate of temperature-change between the lower atmosphere and the tops of the clouds stayed constant. This would be a positive feedback to a postulated warming trend. It seems then that firm predictions based on CO2 air pollution cannot be made. Even the continuation of the rising CO2 concentration trend is in doubt. 4. Albedo and Aerosol
The albedo of a surface is the proportion of energy reflected from it.
The Earth's mean albedo is 0.33 and the mean surface albedo is 0.154
(Otterman, 1977). Much variation exists between different terrains.
There is also much discrepancy between the values given by different
researchers! Henderson-Sellers (1980) for example present surface
albedo values by 3 researchers all in mutual disagreement and none
coinciding fully with the values in table 4.
Table 4
Percentage of energy reflected from various surfaces.
After Table 20.2 in Ramsey et al (1979).
Charney (1975) argued that albedo changes are a factor in
desertification since increased albedo causes cooling hence sinking
atmospheric motion which in turn reduces precipitation.
Possible confirmation of climatic effects from albedo change was presented by Henderson Sellers (1980). Albedo values were measured by satellite-borne radiometers, and British Isles weighted averages for the two dates 20/8/1976 and 28/5/1977, were computed at 0.24 and 0.14 respectively. This variation was above the noise level of seasonal fluctuation, subjective mapping methods and atmospheric absorption changes. The albedo change was suggested as a compounding factor in the North European drought of 5/1975 8/1976. Walker and Rowntree (1977) showed that soil moisture is as important a parameter of rainfall as albedo. Shukla and Mintz (1982) also related soil moisture to precipitation. In other words, not increased albedo alone but increased albedo plus reduced soil moisture act together to promote long term droughts. Sud and Fennessy (1982) confirmed from a model climate study that increased sub-tropic surface albedo promotes overhead atmospheric cooling, sinking air, reduced convection and hence reduced precipitation. The reduced precipitation constitutes positive feedback on drought production. The Mount St. Helens eruption in 1980 temporarily doubled the albedo over Montana and Wyoming (Gribbin, 1982a). The dust reached only an altitude of 24km and had at most a temporary cooling effect. The dust acted as nuclei and promoted rainfall. The extra cloud formation hindered the flow of Westerly winds and this in turn resulted in extremes of weather. Otterman (1977) showed that dust storms do not affect the albedo of a desert. But when the dust is carried over the ocean there is a large albeit temporary and local albedo increase. Humans change Earth's albedo by deforestation, farming, and laying concrete and asphalt. This is not "air" pollution of course. But in the 1970s man ejected up to 400 million tons of aerosol into the atmosphere (Budyko 1977). The result is decreasing atmospheric transparency (McCormick and Ludwig 1967). Aerosols include dust, water droplets, minerals and gases. Agricultural burning, waste burning at oil wells, fossil fuel burning, nuclear tests, industrial emissions and incidents like the "dust bowl" disaster contribute to atmospheric aerosol. Aerosols in the Arctic cannot be accounted for by local pollution, which suggests their transport by winds across thousands of kilometres from industrial regions (Raatz, 1982). Tropospheric dust is washed out by rain and hence has little effect on albedo and climate. But stratospheric aerosol at 20km is above the main weather layer. Aerosols spread quickly over the hemisphere of their origin but comparatively slowly over the other hemisphere. The particles gradually settle under gravitational influence into the troposphere and are washed out. Particles at 20 30km altitude stay an average of about 20 months (Budyko, 1977). Budyko also claimed that aerosol had decreased clear sky direct radiation by 6% in the 1960s. But then the radiation decrease ceased which possibly indicated a stabilization of the aerosol mass (pp. 190-193). In the troposphere the direction of aerosol caused temperature-change is determined by the ratio of the aerosol albedo and the albedo of Earth's surface. If the first is greater temperatures will decrease, if the second is greater temperatures will increase. The mean effective aerosol albedo is 0.42. Comparison with Earth's surface albedo under cloudless conditions gave the conclusion that aerosol decreased temperatures of ice free and snow free surfaces but increased temperatures over ice or snow covered surfaces. Since ice and snow cover less than 20% of Earth's land surface and since the Polar regions get less solar radiation anyway, the net aerosol effect in cloud free areas is to decrease the temperature. In cloudy areas the aerosol effect on the thermal regime is minimal. This is because most tropospheric aerosol lies below the upper cloud boundary (Budyko, 1977). Since global cloud cover averages about 31%, aerosols have a net global cooling effect. Aerosols also, however, block infrared radiation but the albedo effect is greater than this also (Gilliland, 1982). Aerosol may effect not only the atmospheric albedo but also the surface albedo after it settles. Global ice and snow cover averages about 35,000,000km2. But it varies — the difference between 1971 and 1972 being about 4,000,000km2. Bolin (1977) presented a graph relating changes in Northern Hemisphere albedo to changes in ice and snow cover. Energy reflected due to albedo-change varied from 1.75 x 1019 to 2.70 x 1019 calories/day. Warren and Wiscombe (1980) accounted for wide variations in snow albedo measurements by different researchers, by contamination of the Arctic and Antarctic with desert dust and carbon soot. This altered the snow's visible spectrum albedo and led to energy absorption. Thus carbon soot aerosol may continue to exert a warming effect on climate even after its removal from the atmosphere. This effect was not allowed for in the investigations by Budyko and others who compared the aerosol cooling and warming effects. 5. Clouds
Average global cloud cover is about 31%. The largest daily variations
in Earth's albedo result from variations in cloud cover which air
pollution may be affecting.
Taylor (1972) cited an estimate that 1,500 jet aircraft were daily making contrails of 0.5km width, increasing the cover of cirrus clouds over North America and Europe by 5-10%. Newell (1970) showed that the vapour remained in the stratosphere for years. Twomey (1980) reported on cloud condensation nuclei (CCN) on which droplets form in cooling air. Most CCN form from reactions of sulphur-containing trace gases. The CCN do not fall to Earth's surface as rapidly as particulates and hence will rise in concentration over a much greater volume of the atmosphere. Charlock and Sellers (1980) showed that increasing CCN concentration could alter the quantity and duration of cloud cover and hence the frequency and quantity of precipitation. Manabe and Wetherald (1967) estimated that a change in global low cloud cover of 1% would change temperatures by 0.8oC. Hartman and Short (1980) demonstrated that the statistics of the Earth's radiation budget measurements, planetary albedo, and outgoing radiation are all related to cloudiness and to the atmospheric general circulation. High clouds, more than low clouds, retard infrared radiation. Geographical patterns of infrared and albedo were related to air circulation, precipitation zones, deserts and storm zones. Previous researchers (for example Cess, 1976; Ohring and Clapp, 1980) had much debated the relative heating and cooling effects. Cess (1976) argued that even if the feedback effects did cancel, this would not be the same seasonally or zonally. Hartman and Short's conclusion on the global effect of cloud albedo on the radiation budget was that the heat-loss through cloud reflection is 2.5 times the infrared retained. Harshvardhan (1982) pointed out that model-studies of clouds, usually presumed fixed cloudiness and simple cloud configurations. There was no allowance for brokenness. Yet half of all tropical cloudiness and 1/3 of sub-tropical cloudiness is classified as "broken". The different conditions of broken cloud can alter the magnitude of the feedback effects as much as three fold. The Hartman and Short investigation apparently overcame this problem through the use of actual measurements by satellite-borne instruments to evaluate the net radiative effect of present cloud distributions. There were no assumptions regarding cloud albedo, cloud height or area of cloud coverage. The calculation of the radiation budget was based only on measurements of planetary albedo and outgoing terrestrial radiation. There are further complexities. Most climate models assume fixed optical properties for each cloud type. Charlock (1982) showed that optical properties of clouds in both solar and infrared wavelengths are related to the cloud's liquid water content (LWC). Temperature variation is sensitively tuned to variable cloud optics particularly cirrus clouds. Recent cloud climatologies are able to assess the amount of low cloud to an accuracy of 0.05 of sky cover. But because of variable optics a 0.05 error could represent a 3oC temperature difference! Another determinant of cloud optics is the droplet distribution, which in turn is subject to CCN concentrations (see above) in turn related to air pollution. The effect on the radiation balance of cloud optics also varies with season and latitude (Potter et al 1981). Generally the effect of cloud-optics constitute negative feedback on climate. However, a climatically induced vertical redistribution of LWC would in turn alter cloud optics even if total LWC remained constant. These are new areas of research and until resolved they cast doubt on the previous conclusion that a cloud build-up would have a net cooling effect. AIR POLLUTION and THE WORLD'S CLIMATE
PART 2 Anonymous (Investigator #198, 2021 May)
6. Chlorinated Compounds and Ozone (O3)
CF2Cl2 and CFCl3 are chlorofluorocarbons (CFCs) i.e. compounds of chlorine, fluorine and carbon (Perner (1979).
CFCs absorb both ultraviolet (UV) and infrared radiation, thereby warming the atmosphere by slowing the loss of infrared radiation to Space. If CFC concentration in the atmosphere increases the result would be a further greenhouse effect. Ramathan (1975) using a, radiative convective model computed a 0.9oC temperature increase by the year 2000 when CF2Cl2 and CFCl3 reached concentrations of 2ppb. CFCs have a further effect. They decompose in the atmosphere giving the decomposition product chlorine. Chlorine destroys ozone (O3). Ozone is a poisonous form of oxygen which also has a greenhouse effect because it absorbs UV radiation, but will decrease 15% when the above concentration (2ppb) of CFCs is reached. Thus CFC pollution itself adds to the greenhouse effect, but the chlorine that it produces destroys O3 — a climate cooling effect. O3 absorbs much of the Sun's dangerous incoming UV radiation as well as infrared. A 15% decline in O3 levels would increase the UV radiation reaching Earth's surface 30%. This would decimate plant and animal life and the decreased plant cover would increase Earth's albedo (cooling effect). But less plant-cover implies decreased photosynthesis hence increased CO2 levels (warming effect). 2% of all incoming solar energy is absorbed by O3 and is responsible for the stratospheric thermal structure. Vertical and latitudinal temperature gradient changes would affect air movements and winds (Ramathan et al, 1976). Fels et al (1980) calculated that a 50% decrease of O3 would weaken the mid-latitude Westerlies and reduce stratospheric heating. Callis and Natarajan (1981) studied the combined effects of CO2 and CFCs, using already existing radiative convective models. Increased surface temperature led to increased tropospheric water vapour which affected tropospheric photochemistry leading to OH production which in turn raised HO2 levels. HO2 catalytic action promoted additional ozone destruction which levelled off at 5.5% reduction after 50 years. Here we need to discuss another pollutant, nitrogen oxides (NOx), which may also change ozone levels.
7. Nitrogen and Ozone
Air is 78% by volume nitrogen gas (N2) which moves between atmosphere, soil, water and living things in a continuous cycle.
Biological reactions in soil "fix" (i.e. incorporate into compounds) nitrogen gas from the atmosphere into ammonium (NH4), which bacteria together with oxygen convert to nitrites (compounds with one nitrogen atom combined with two oxygen atoms, NO2), and then to nitrates (compounds with one nitrogen atom combined with three oxygen atoms, NO3). Nitrates are what plants assimilate. When plants die and decay, nitrates get converted back to nitrogen and nitrous oxide (N2O), which reach the stratosphere by diffusion and convection. There the N2O decomposes to nitric oxide (NO) which reacts with ozone (O3) converting it to oxygen (O2). [Details of the nitrogen cycle are given in Pratt el al (1977) and Hahn (1979)]. The nitrogen cycle therefore destroys ozone — a cooling effect. But increasing N2O levels produce a further greenhouse or warming effect! Ordinarily a natural balance exists between ozone destruction and its formation by the reaction of light with oxygen. But air pollution is now a significant addition to the nitrogen cycle: Of global atmospheric nitrogen "fixed", combustion accounts for 8.4% and industry 4.6% (Pratt et al, 1977). In 1974 nitrogen fertiliser added to farm soils reached 40 million tons. The fertilizer eventually also generates N2O. Combustion in cars and supersonic exhausts from aircraft (Johnston 1971) result in NO directly, and N2 and NO2 indirectly. Thus both combustion and photochemical processes are important in the global ozone cycle (Gidel and Shapiro, 1980). Destruction of ozone via nitrogen oxides could theoretically perturb the natural cycles toward global cooling (because lowered O3 levels imply decreased absorption of UV and infrared radiation). Alternatively, the accumulation of nitrogenous gases, N2O, NH3 and HNO3 could act as a further greenhouse effect (Wang et al, 1976). By the year 2000 this could equal half the CO2 effect (Hahn, 1979). Hameed and Paidoussis(1981) pointed out that from 3 to 60 million tons of tropospheric nitrogen, a controlling factor in the O3 budget, is produced by lightning discharges annually. Liu et al (1980) suggested that the main source of tropospheric O3 is its in situ production via photochemical processes on a continuous small influx of stratospheric oxides of nitrogen, which disagrees with Pratt et all that these oxides decrease ozone levels. Fishman (1981) in turn disagreed (with Liu et all) because the computed NOx distribution in the troposphere and hence the distribution of photochemical O3 production disagree with recent available NOx measurements. A cross tropospheric flux of NOx and other active nitrogen compounds had also been ignored. Also, anthropogenic NOx emissions are 100 times greater than the stratospheric NOx influx. Furthermore, 75% of the Northern Hemisphere-produced tropospheric O3 is produced below 45o latitude and below 5km altitude. At 2km and 9km altitudes the NOx concentrations are almost equal, yet the O3 production rate is 4 times greater at 2km, possibly because other O3 precursors such as carbon monoxide (CO) and CH4 are more abundant at 2km. In short, "Liu et all ... on the origin of tropospheric ozone may not be correct." (p. 12,163) Hameed and Stewart (1983) summarized previous analyses and models of the O3 budget. Their own model's successful reproduction of latitudinal O3 variation depended on the inclusion of photochemical processes. The authors pointed out that photochemical reactions of non-methane hydrocarbons and the effect of thunderstorms on the distribution and magnitude of NOx are both poorly researched. The troposphere contains 10% of all O3. Because this 10% is under greater pressure than stratospheric O3 it absorbs infrared to an extent equal to the latter and exerts 50% of the warming effect of O3. The altitude at which O3 formation and destruction occur is therefore relevant but earlier researchers ignored this aspect. Carbon monoxide (CO) is another emission of fossil fuel burning. It combines with oxygen to produced carbon dioxide. It also reacts with chemicals that destroy ozone and methane. For example CO converts OH to HO2 which in turn converts the ozone-destroying gas NO to NO2. (Bell, 1982; Khalil and Rasmussen, 1983; Hameed et al, 1980). Methane (CH4) is an air pollutant released by gas fields, coal mines and rice fields. From 1970 to 1980 the atmospheric concentration of methane increased from 1,400ppb to 1,600ppb (Craig and Chou, 1982; Rasmussen and Khalil, 1981). CH4 converts atmospheric chlorine to hydrochloric acid which in turn would apparently reduce the ozone-destroying effect of chlorine (produced by the previously discussed CFCs). The concentration of CH4 is, however, itself decreased by reaction with the hydroxyl ion OH. CH4 oxidation moreover produces O3 and CO. This still leaves us with the CH4 and O3 greenhouse effects except that O3 increase is not clearly demonstrated. The sources, sinks and atmospheric concentrations of some of the other chemicals are also debated. N2O together with methane (CH4) could add 2oC to mean surface temperatures (Donner and Ramathan, 1980). But O3 depletion may be a prior danger, since a 7% decrease would expose humans to dangerous UV levels. Hahn (1979) forecast a depletion of 3% to 13% by 2050 AD. (p. 210) Reinsel et al (1982), however, found data on ozone trends to be inconclusive. Conclusion: No definitive decision regarding the cumulative, overall impact on climate of these chemicals is yet possible.
8. Thermal Pollution
Kellogg and Schware (1981) reveal that the average world annual growth-rate in human energy consumption is 5.6% (1960-1970 and 3.2% (1970-1978).
Table 5
Past energy consumption
Extracted from Table A.1 in Kellogg and Schware (1981)
Bolin (1977) gives the global 1970 values of energy flux per unit area
as 0.016 W/m2 for human energy production, and as 240 W/m2 for the
average solar energy received by the Earth and radiated back to Space.
The 240 W/m2 is fairly constant. Budyko (1977) suggests that if the radiation balance increased by 0.2 - 0.6 W/m2 the polar ice would start to melt. Projecting the 3.2% annual growth in anthropogenic energy consumption (energy handled by man and ultimately converted to heat) for eighty years would result in 1.03280 x 0.016 = 0.2 W/m2 which is the lower estimate for when the polar ice melts. The chief sources of such heat would be nuclear power station cooling, industrial heat, and heat generated by the activities of an ever-increasing human population. Fossil fuel use cannot by itself produce the projected energy increase (Bolin, 1977; Krenz, 1977); nuclear energy use would be necessary. Already there are "heat islands" such as New York, Chicago and Moscow, which are significantly warmer than surrounding areas. Bolin (1977) reported that such cities experience decreased temperature differences between day and night, less effective solar radiation due to reflection, and increased haze, fog and precipitation. There is also more turbidity and a longer growing season than in surrounding areas. Washington (1972) related thermal pollution to population density and discovered no climatic effect. Damen and Kunen (1976), however, demonstrated a slight (0.2oC) warming in five of eight Southern Hemisphere cities with populations exceeding 750,000. The confidence level was 92.5%. Smaller cities showed no heating effect. Egger (1979) challenged previous conclusions regarding impact on climate from thermal pollution, and sees only a weak affect on atmospheric circulation. "Heat islands", where local climates have indeed changed do not have temperature build-ups to intolerable levels because the heat is distributed laterally as well as vertically. But what if thermal pollution on the New York scale becomes a global phenomenon, leaving little space for lateral heat distribution to take place? Also, a 12.5 fold increase in thermal pollution reaches the lower estimate (12.5 x 0.016 + 240 = 240.2 W/m2) at which polar ice starts to melt.
9. Further Considerations
Pollution in the air interacts with ground-level pollution and other
events. Oil films on water affect the oceanic-atmospheric heat
exchange. Partial melting of polar ice would provide a layer of less
dense surface water which may affect the heat exchange and CO2
exchange. This could in turn change the ocean's role as a thermal
buffer.
Irrigation, artificial lakes and reservoirs may alter the balance between evaporation and precipitation. Water evaporating takes heat with it but thereafter increases infrared absorption. The comparative evaporative rates of soil, forests, fields and oceans are not well researched. Differential heating, especially the contrast between equator and poles, is important. A higher surface temperature would speed up air circulation patterns. Revelle (1982) made reference to a mathematical model to explore geographical distributions of climatic changes resulting from CO2 doubling. Tropical temperature rose 2oC, at 35o north 3oC, at 50o north 4oC, and within the Arctic Circle 7oC. The model made no allowance for cloud response and albedo, and handled atmospheric/oceanic heat exchange inadequately. One would also expect the prescribed temperature changes to alter the ocean currents. 70% of photosynthesis occurs in the oceans, but ocean pollution may alter the species composition of oxygen-producing algae. If oceanic production of oxygen thereby decreased, raising atmospheric CO2 concentration, this might be balanced by the extra CO2 stimulating the oxygen-producing algae! Several esoteric possibilities already under discussion may further increase global photosynthesis. Icebergs towed to dry areas may provide water to greenify the deserts. Artificial "suns" consisting of light reflected by orbiting space platforms may extend growing seasons. Another circumstance, potentially so important as to place qualifications on everything discussed hitherto, is the possible instability of the West Antarctic Ice Sheet. An area of 3 million km2 is held in place by submerged mountain ranges, some at 1km below sea level. Instability is evidenced by the generally concave surface, basal melting and decreasing glacial area (Hughes, 1973). Changes in the ice volume, if for example temperatures rose 4oC, could lead to surges in the ice followed by disintegration and collapse into the surrounding oceans. Schneider and Chen (1980) estimated that a potential 7.6 metre ocean level rise would inundate 2.1% of the USA land area ousting 16 million people. If the collapse occurred quickly, say 1-week, the destruction of coastal cities and harbours by tsunamis could be comparable to damage wrought by a nuclear war. Glaciologists disagree on the probability of this event (Weertman, 1976), but study it seriously because of the potential for coastal destruction and climatic changes. Some (see Schneider and Chen, 1980) argue that the current ice profile is typical of a slow build up post-surge state and that the ice mass is thickening, not thinning. Others, e.g. Budd and McInnes (1975) showed that large ice surges are at present physically possible. The relevance of air pollution on the Antarctic ice lies not only in the potential greenhouse and aerosol/cloud albedo effects on Antarctic precipitation. The settling of dust and carbon soot from industrial areas changes the ice-albedo, increasing energy absorption (see above under "Albedo and Aerosol") perhaps adding to any ice thinning trend. Assuming a slow ice sheet disintegration (e.g. 1 generation), how are other climate influencing factors affected? Ocean surface waters would have lower salinity and also a greater area. Infrared absorption, CO2 absorption and evaporation rates would alter. Reduced land and ice areas would be a positive feedback because of the lower albedo of water. We can also imagine a desperate scramble by humans to relocate industry and other resources to higher ground and in the process increasing fossil fuel use and air pollution. Reduced land area would also reduce forest and other plant cover. This scenario of ice sheet disintegration is speculative since experts disagree on the instability question. Oerlemans (1982b) in contrast concludes that global warming would expand the Antarctic ice sheet! Whether the increase to Earth's surface albedo caused by the greater sea ice area could counter a greenhouse effect is another unanswered question. Several times above I mentioned forests and vegetation. How do forests relate to air pollution and climate? Consider air pollutants that lead to acid rain: Sulphur dioxide released by burning of fossil fuel (coal & oil) combine with water vapour to form sulphurous acid — SO2+H2O à H2SO3 — an intermediate product to acid rain. Sulphur trioxide (SO3) with water forms sulphuric acid — SO3+H2O à H2SO4 the primary cause of acid rain. Sulphur oxides, however, at current pollution levels rapidly disperse and have an atmospheric residence time of only 3 to 8 days (Georgii, 1979). Another source of acid rain is NOx (nitrogen oxides from fossil fuel burning) reacting with water and oxygen to form nitric acids. Rain in North America and Europe is also laden with toxic heavy metal pollution released by industry. In addition, increased local O3 levels may open leaf-pores facilitating leaching of nutrients by acid rain. The polluted rain may be at least partly responsible for stunted growth and deaths affecting a third of the trees in woodlands of West Germany and some American states (Toufexis, 1984). Another series of air pollutants, released by the combustion of gasoline and oil, are the hydrocarbons including paraffins, olefines, acetylines, benzenes, naphthenes and anthrathenes. These react with sunlight causing photochemical pollution, which adds to the damage done to plants. Global natural emissions are 768 million tons annually, and by humans (in 1970) 53 million tons (Greenwood and Edwards, 1973). Forests damaged by air-pollution, aggravated by deliberate clearing, changes the CO2/O2/biomass/photosynthesis relationships. Forests also decrease mean wind speeds hence turbulence; air streams tend to arch over forests and this rising promotes precipitation. This changes if air pollution reduces global forest cover. When forest-cover declines local humidity decreases and may decrease below condensation level due to intensified surface solar heating — but albedo would increase, decreasing the local heating (Budyko, 1977). The net effect to global heating or cooling of all these considerations is uncertain.
10. Crazy weather
A warming trend started after WWII seemingly confirming the CO2
greenhouse effect theory. In 1970 the mean global surface temperature
was 0.3oC higher than in 1940 (Budyko, 1977; Flohn, 1977). Then
followed a cooling trend, equally small, apparently confirming the
effects of increased albedo.
Newspaper reports about "Crazy weather is world-wide" (e.g. Sunday Mail, January 7, 1973, p. 27) were causing some alarm. Sweden experienced its warmest winter in 200 years in 1972/73. A new equatorial dry belt was blamed for the drought in East Africa and for decreased Monsoonal rain in India. In Siberia thermometers were falling lower and in Canada 8 inches of snow fell on the western wheat fields — in summer! The USA experienced two consecutive very cold winters 1976/77, 1977/78 (Chico and Sellers, 1979), yet in Germany during Christmas 1974 people enjoyed balmy 20oC warmth. Figure 2, a copy of the article "Crazy weather is world-wide", is here omitted
Before we jump to conclusions, one further set of factors must be considered. From the relative abundance of two types of oxygen in a 139-metre Greenland ice-core representing 100,000 years, past temperatures at the times of precipitation were calculated (Dansgaard, 1969). Rapid and gradual past temperature shifts had occurred. A 13,000-year cycle correlated with oscillations in the Earth's axis, and cycles of 120 and 940 years appeared related to changes in solar radiation. Gribbin (1983) pointed to a 76-year cycle in changes of the sun's diameter. Whenever minimum diameter coincided with maximum sunspot activity (an 11 year cycle) the sun's luminosity could decrease significantly. Gilliland (1982) combined CO2 trends, volcanic activity and a 0.28% solar luminosity change during a 76-year cycle in a climatic model and got a close match with actual records. The pre-WWII warm era was accounted for by the sun being at maximum luminosity and volcanic eruptions being few. Another extraterrestrial climatic influence may occur when most of the planets bunch up on one side of the sun as in 1982, 1844 and 1665. Their gravitational force increases the radius of Earth's orbit 1½ million miles. This should reduce the radiation reaching Earth, but in 1982 there was no measurable effect. Gribbin (1982b) pointed out the speed of the orbit also changes during such a "synod". When Earth is on the side of the sun opposite to the bunched-up planets its orbital speed increases; when on the same side the gravitational pull of the planets slows it down. This alters the length of the seasons on Earth. The 1665 synod coincided with the coldest decades of the "little ice age".
11. Conclusions
It would have been encouraging to complete this review with something as plain as: "If we pollute the atmosphere in manner w at rate x, the climatic affect in y years will be z." But the physical basis of climate is not that simple. We cannot even be sure that most of the relevant influences have been mentioned. Past temperature variability, past volcanic variability, past CO2 variability, past solar-luminosity variability, and past climatic fluctuations such as the "mini ice age" (1400 to 1700 AD), constitute evidence that the total effect of all air pollution is but "noise" in the major determinants of climate. The research of Dool et al (1978), Chico and Sellers (1979) and others, suggests that climatic variability even in recent generations, exceeded the conditions of 1931 1960. And of course the ice ages of the more distant past are proof of extreme climatic swings when man had no influence on climate at all. If we define normal climate as being the conditions of 1931-1960 and blame departures from those conditions on air pollution we have merely made our conclusion true by definition. What we have to know before we can reasonably impugn air pollution is how the world's climate would swing irrespective of pollution. Comparing the so-called "crazy weather" of the 1970s to a spinning top (as some have done) which gyrates more erratically as it slows down and loses stability, goes beyond the evidence. Even if we accept the definition of "normal climate", no parameter has passed the noise level. Table 6 summarises the theoretical effect on global temperatures of air pollutants. The table includes major factors such as ice caps and forests which are affected by air pollution and which in turn influence global temperatures (theoretically if not measurably).
Table 6
Affect on global temperatures when various factors are increased
(in concentration, rate, size, area or number as appropriate)
Most air pollutants seem either to have a mutual cancelling effect with respect to their influence on temperatures or they create other conditions which influence temperatures in the direction opposite to the pollutant. The chlorofluoromethanes and the nitrogen compounds, for example, may have a cancelling effect. Acid rain destroys forests, decreasing photosynthesis, enhancing the CO2 effect which in turn may be partly negated (as far as temperatures are concerned) by increased surface reflectivity. Thermal pollution seems potentially the most imminent threat. Projecting the 3.2% annual increase in human energy consumption, as calculated above, gives 80 years to reach the lower level of global heating at which Budyko suggests the polar ice starts melting. The thermal pollution would also increase the atmospheric water vapour which is a positive feedback due to infrared retention, but the further interactions of the water vapour with clouds, atmospheric albedo and atmospheric chemicals render the net temperature effect less plain. Raised surface temperature (of say 2oC) would also lead to the expansion of the Antarctic ice sheet (currently 15 million km2). The world's ice free or seasonally ice free ocean area (about 280 million km2) would correspondingly decrease. Since the albedo of ice and snow is 20 times that of water the extra heat loss by solar energy reflection would surely make a difference. I am not denying that climatic belts have shifted or that average global temperature and precipitation have changed. But I am questioning whether air pollution is the main cause. This century's so called "warm" and "cold" periods differed by only 0.3oC. The 0.05 error in cloud cover computations represents 3oC. In other words non measurable natural fluctuations in cloud cover alone, might account for so called "temperature changes" that are 10 times greater than the recent global change. What we require are accurate measures of all relevant climate affecting factors, and improved models which incorporate all relevant feedback and interactive factors. The latest models incorporate knowledge of radiation transfer, vertical energy transport, surface/atmospheric thermodynamic interaction, and albedo effects. Much greater sophistication, however, will be needed. If the climatic effects of future air pollution pass the level of noise, at least research may have revealed the remedial actions that have least ramifications. CO2 warming for example could be countered by planting an extra billion trees per year or by blocking solar radiation with dust raised with nuclear explosions. Already, in this instance we know that the former solution is better. It may also be the case (as with the potential ozone loss) that air pollution to the extent of altering global climate would be lethal to mankind first! Clearly, the question of air pollution and world climate needs continued monitoring.
REFERENCES
BELL, A. (1982) Methane, too, may be warming the atmosphere. Ecos, 33, 8-10 BERNER, W., OESCHGER, H, & STAUFFER, B. (1980) Information on the CO2 Cycle from Ice Core Studies, Radiocarbon, Volume 22, No. 2, 227-235 BOLIN, B. (1977) The impact of production and use of energy on the global climate. Ann. Rev. Energy, 2, 197-226 BUDD, W.F. & McINNES, E. (1975) Modelling periodically surging glaciers, Science, 186, 925-927 BUDYKO, M.I. (1977) Climatic Changes, Waverly Press (Maryland) CALLENDER, G.S. (1938) The artificial production of carbon dioxide and its influence on temperature. Quart. J. Roy. Met. Soc., 64, 223-240 CALLIS, L.B. & NATARAJAN, M. (1981) Atmospheric carbon dioxide and chlorofluoromethanes: combined effects on stratospheric ozone, temperature, and surface temperature. Geophysical Research Letters, 8, 587-590 CESS, R.D. (1976) Climatic change: An appraisal of atmospheric feedback mechanisms employing zonal climatology, J. Atmos. Sci., 33 1831-1843 CESS, R.D. & GOLDENBERG, S.D. (1981) The effect of ocean heat capacity upon global warming due to increasing atmospheric carbon dioxide, J. Geophys. Res., 86, 498-502 CHALMERS, A.F. (1976) What is this thing called Science? University of Queensland Press (Queensland) CHARLOCK, T.P. (1982) Cloud optical feedback and climate stability in a radiative convective model., Tellus, 34, 245-254 CHARLOCK, T.P. & SELLERS, W.D. (1980) Aerosol cloud reflectivity and climate. J. Atmos. Sci., 37, 1136-1137 CHARNEY, J.G. (1975) Dynamics of deserts and droughts in the Sahel, Quart. J. Roy. Met. Soc., 101, 193-202 CHERVIN, R.M. (1980) On the simulation of climate and climatic change with general circulation models. J. Atmos. Sci., 37, 1903-1913 CHICO, T. & SELLERS, W.D. (1979) International temperature variability in the United States since 1896, Climatic Change, 2, 139-147 CRAIG, H. & CHOU, C.C. (1982) Methane: the record in polar cores, Geophysical Research Letters, 9, 1221-1224 DAMIEN, P.E. & KUNEN, S.M. (1976) Global Cooling? Science, 193, 447-453 DANSGAARD, W., JOHNSEN, S.J., MOLLER, J. & LANGWAY, C.G. (1969) One thousand centuries of climatic record from Camp Century on the Greenland Ice Sheet, Science, 166, 377-381 DEEVEY, E.S. (1958) Bogs, Sci. Amer. 199, 114-122 DELMAS, R.J., ASCENCIO, J.M. & LEGRAND, M. (1980) Polar ice evidence that atmospheric CO2 20000 yr. BP was 50% of present, Nature, 284, 155 DONNER, L. & RAMANATHAN, V. (1980) Methane and nitrous oxide: Their effects on terrestrial climate. J. Atmos. Sci., 37, 119-124 DOOL, H.M. VAN DEN, KRIJNEN, H.J. & SCHNURMANS, G.J.E. (1978) Average winter temperatures at De Bilt (The Netherlands 1634 1977, Climatic Change, 1, 319-330 DORF, E. (1960) Climatic changes of the past and present, American Scientist, 48, 341-364 EGGER, J. (1979) The impact of waste heat on atmospheric circulation, Developments in Atmospheric Science, Vol. 10, Elsevier, Amsterdam. FELS, S.B., MAHLMAN, J.D., SCHWARZKOPF, M.D. & SINCLAIR, R.W. (1980) Stratospheric sensitivity to perturbations in ozone and carbon dioxide: radiative and dynamic response, J. Atmos. Sci. 37, 2265-2297 FISHMAN, J. (1981) The distribution of NOx and the production of ozone: Comment on 'On the origin of tropospheric ozone,' by S.C. Liu et al. J. Geophys. Res. 86, 12161-12164 FLOHN, H. (1977) Climate and energy, Climatic Change, 1, 5-20 GEORGII, H.W. (1979) Large scale distribution of gaseous and particulate sulphur compounds and its impact on climate, Developments in Atmospheric Science, Vol. 10, Elsevier, Amsterdam GIDEL, L.T. & SHAPIRO, M.A. (1980) General circulation estimates of the net vertical flux of ozone in the lower stratosphere and the implications for the tropospheric ozone budget. J. Geophys. Res. 85, 4049-4058 GILLILAND, R.L. (1982) Solar, volcanic, and CO2 forcing of recent climatic changes, Climatic Change, 4, 111-131 GREENWOOD, N.H. & EDWARDS, J.M.B. (1973) Human Environments and Natural Systems. Duxbury (Britain) GRIBBIN, J. (1982a) Do volcanoes affect climate? New Scientist, 93, 150-153 GRIBBIN, J. (1982b) Stand by for bad winters, New Scientist, 96, 220-223 GRIBBIN, J. (1983) The curious case of the shrinking sun, New Scientist, 97, 592-595 HAHN, J. (1979) Man made perturbation of the nitrogen cycle and its possible impact on climate. In: Bach, W., Pankrath, J. and Kellogg, W. (Editors) Developments in Atmospheric Science, Vol. 10, 193-213, Elsevier, Amsterdam Hall, D.C. (1979) Biological solar energy conversion for fuels, Nature, 278, 114-117 HAMEED, S., CESS, R.D. & HOGAN, J.S. (1980) Response of the global climate to changes in atmospheric chemical composition due to fossil fuel burning, J. Geophys. Res. 85, 7537-7545 HAMEED, S. & PAIDOUSSIS, O.G. (1981) Implications of natural sources for the latitudinal gradients of NOy in the polluted troposphere, Geophysical Research Letters, 8, 591-594 HAMEED, S. & STEWART, R.W. (1983) Latitudinal variation of tropospheric ozone in a photochemical model, J. Geophys. Res. 88 5153-5167 HARDIN, G. (1961) Biology: Its Principles and Applications, Freeman HARSHVARDHAN (1982) The effect of brokenness on cloud-climate sensitivity. J. Atmos. Sci. 39, 1853-1861 HARTLINE, B.K. (1980) Coastal upwelling: physical factors feed fish, Science 208, 38-40 HARTMAN, D.L. & SHORT, D.A. (1980) On the use of earth radiation budget statistics for studies of clouds and climate. J. Atmos. Sci. 37, 1233-1250 HENDERSON-SELLERS, A. (1980) Albedo changes – surface surveillance from satellites, Climatic Change 2, 275 81 HOFFERT, M.I., CALLEGARI, A.J. & HSIEH, C.-T. (1980) The role of deep sea heat storage in the secular response to climate forcing, J. Geophys. Res. 85, 6667-6679 HOYT, D.V. (1979) Variations in sunspot structure and climate, Climatic Change 2, 79-92 HUGHES, T. (1973) Is the West Antarctic Icesheet disintegrating? J. Geophys. Res. 78 7884-7910 JOHNSTON, H. (1971) Reduction of stratospheric ozone by nitrogen oxide catalysts from supersonic transport exhaust, Science 173, 517- 522 KEELING, C.D. (1978) Atmospheric carbon dioxide in the 19th century, Science 202, 1109 KELLOGG, W. & SCHWARE, R. (1981) Climatic change and society: consequences of increasing atmospheric carbon dioxide, Westview KHALIL, M.A.K. & RASMUSSEN, R.A. (1983) Sources, sinks and seasonal cycles of atmospheric methane, J. Geophys. Res. 88, 5131-5144 KRENZ, J.H. (1977) Minimizing the environmental impact of more effective energy utilization, Climatic Change 1, 307-318 LIU, S.C., KLEY, D., McFARLAND, M., MAHLMAN, J.D. & LEVY, H. (1980) On the origin of tropospheric ozone, J. Geophys. Res. 85, 7546-7552 MANABE, S., BRYAN, K. & SPELMAN, M.J. (1975) A global ocean atmosphere climate model, Part 1, The atmospheric circulation. J. Oceanogr. 5, 3-29 MANABE, S. & STOUFFER, R. (1979) Study of climatic impact of CO2 increase with a mathematical model of global climate, Nature 282, 491-493 MANABE, S. & WETHERALD, R.T. (1967) Thermal equilibrium of the atmosphere with a given distribution of relative humidity, J. Atmos. Sci. 24, 241-259 MANABE, S. & WETHERALD, R.T. (1980) On the distribution of climatic change resulting from an increase in CO2 content of the atmosphere., J. Atmos. Sci. 37, 99-118 MANABE, S., WETHERALD, H.T. & STOUFFER, R.J. (1981) Summer dryness due to an increase of atmospheric CO2 concentration. Climatic Change 3: 347-385 McCORMIC, R.A. & LUDWIG, J.H. (1967) Climate modification by atmospheric aerosols, Science 156, 1358-1359 MOLINA, M.J. & ROWLAND, F.S. (1974) Stratospheric sink for chlorofluoromethanes: chlorine atomic catalysed destruction of ozone, Nature 249: 810-812 NEWELL, R.E. (1970) Water vapour pollution in the stratosphere by the supersonic transporter? Nature 226: 71 OERLEMANS, J. (1982a) Effect of irregular fluctuations in Antarctic precipitation on global sea level. Nature 290: 770-772 OERLEMANS, J. (1982b) Response of the Antarctic Ice Sheet to a climatic warming: a model study. J. Climatology, 2: 1-11 OHRING, G. & CLAPP, P. (1980) The effects of changes in cloud amount on the net radiation at the top of the atmosphere. Atmos. Sci. 37: 447-454 OTTERMAN, O. (1977) Anthropogenic impact on the albedo of the earth, Climatic Change 1: 137-155 PEARMAN, G.I. & HYSON, P. (1981) The annual variation of Atmospheric CO2 concentration observed in the northern Hemisphere. J. Geophys. Res. 86: 9839-9843 PERNER, D. (1979) The consequences of increasing CFM concentrations for chemical reactions in the stratosphere and their impact on climate, Developments in Atmospheric Science, Vol. 10, pp 215-228 PITTOCK, A.B. et al (seven scientists) (1981) Human impact on the global atmosphere: implications for Australia, Search, 12: 260-272 PLASS, G. (1956) The carbon dioxide theory of climatic change, Tellus, 8, 140-154 POTTER, G.L., ELLSAESSER, H.W., MACCRACKEN, E.C., & MITCHELL, C.S. (1981) Climatic change and cloud feedback: the possible radiative effects of latitudinal distribution. J. Atmos. Sci. 38, 489-493 PRATT, P.F. et al (eleven scientists) (1977) Effect of increased nitrogen fixation on stratospheric ozone, Climatic Change 1: 109-135 RAATZ, W.E. (1982) On the meteorological characteristics of polluted air masses at Barrow, Alaska. Pure and Applied Geophysics 120, 662-672 RAMATHAN, V. (1975) Greenhouse effect due to chlorofluorocarbons: Climatic implications. Science 190, 50-51 RAMATHAN, V., CALLIS, L.B. & BOUGHNER, R.E. (1976) Sensitivity of surface temperature and atmospheric concentration of ozone and nitrogen dioxide. J. Atmos. Sci. 33: 1092-1112 RAMSEY, W.L., PHILLIPS, G.R. & WATENPAUGH, F.K. (1979) Modern earth science, Holt, Rinehart and Winston (USA) RASMUSSEN, R.A. & KHALIL, M.A.K. (1981) Atmospheric methane (CH4): trends and seasonal cycles, J. Geophys. Res. 86: 9826-9832 RASOOL, S.I. & SCHNEIDER, S.H. (1971) Atmospheric carbon dioxide and effects of large increases on global climate, Science, 173: 138-141 REINSEL, G., TIAO, G.C. & LEWIS, R. (1982) A statistical analysis of total ozone data from the Nimbus 4 BUV satellite experiment, J. Atmos. Sci. 39: 418-443 REVELLE, R. (1982) Carbon dioxide and world climate, Sci. Amer. 247, No. 2, August, 35-43 ROSENBERG, N.J. (1981) The increasing CO2 concentration in the atmosphere and its implication on agricultural productivity II. effects through CO2 induced climatic change, Climatic Change 3: 265-279 ROTTY, R.M. (1979) Energy demand and global climate change, Developments in Atmospheric Science Vol. 10, 269-273 Elsevier, Amsterdam SCHNEIDER, S.H. & CHEN, R.S. (1980) Carbon dioxide warming and coastline flooding, Ann. Rev. Energy, 5: 107-140 SEILER, J. & CRUTZEN, P.J. (1980) Estimate of gross and net fluxes of carbon between the biosphere and the atmosphere from biomass burning, Climatic Change 2: 207-247 SHUKLA, J. & MINTZ, Y. (1982) The influence of land surface evaporation on the earth's climate, Science, 215: 1498-1501 SINGER, S.F. (1970) Human energy production as process in the biosphere, Sci. Amer. 223: 183-186 STRINGER, E.T. (1972) Techniques of Climatology, Freeman (USA) SUD, Y.C. & FENNESSY, M. (1982) A study of the influence of surface albedo on July circulation in semi arid regions using GLAS GCM, J. Climatology, 2: 105-125 SWINBURNE, R. (1974) The justification of induction, Oxford (Great Britain) TAYLOR, G.R. (1972) The Doomsday Book, Panther (London) THOMPSON, K. (1981)The question of climatic stability in America before 1900, Climatic Change 3: 227-241 THOMPSON, S.L. & SCHNEIDER, S.H. (1981) Carbon dioxide and climate: ice and ocean, Nature 290, 9-10 TOUFEXIS, A. (1984) Puzzling holes in the forest, Time, 123, No. 14, 72 TWOMEY, S. (1980) Cloud nucleation in the atmosphere and the influence of nucleus concentration levels in atmospheric physics, J. Phys. Chem. 84: 1459-1463 WALKER, J. & ROWNTREE, P.R. (1977) The effect of soil moisture in a tropical model, Quart. J. Roy. Met. Soc. 103: 29-46 WANG, W.C., YUNG, Y.L., LACIS, A.A., NO, T. & HANSEN, J.E. (1976) Greenhouse effects due to man made perturbations of trace gases, Science, 194: 685-690 WARREN, S.G. & WISCOMBE, W.J. (1980) A model for the spectral albedo of snow. 11: Snow containing atmospheric aerosols, J. Atmos. Sci., 12: 2754-2745 WASHINGTON, W.M. (1972) Numerical climatic change experiments; the effect of man's production of thermal energy, J. Appl. Met. 11: 768-772 WEERTMAN, T. (1976) Glaciology's grand unsolved problem, Nature 260: 284-286 |